
Bioptechs chambers offer an easy-to-use, sophisticated solution for high resolution imaging. With an emphasis on what matters most to your lab.
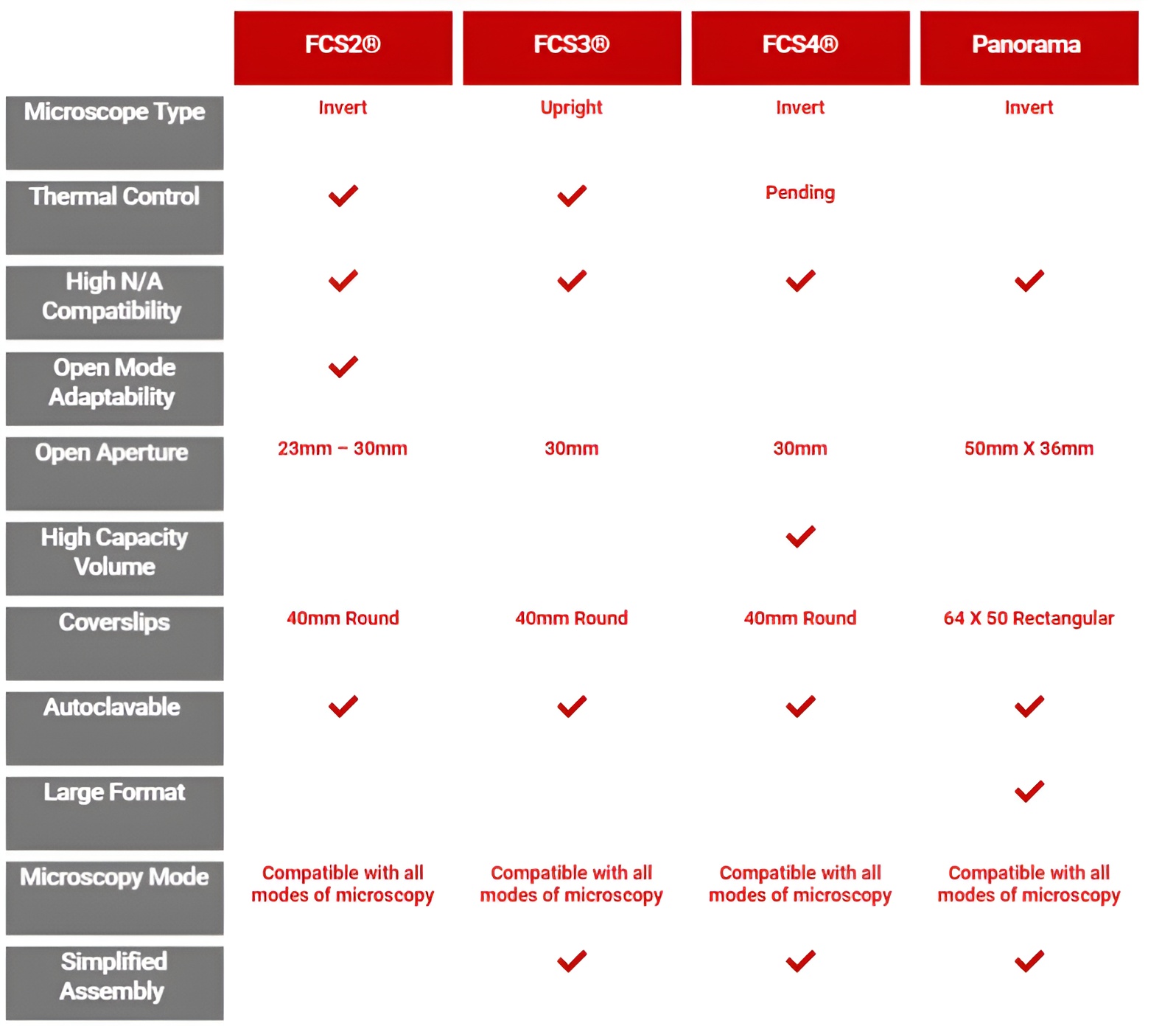
The FCS2 Chamber System the most precise micro-environmental control system on the market. The FCS2 is a parallel plate, high N.A. compatible, uniformly temperature controlled microscopic flow cell with a user definable specimen area for inverted microscopes. Intelligent feedback thermal loop prevents overshoot on temperature and protects the integrity of the sample.
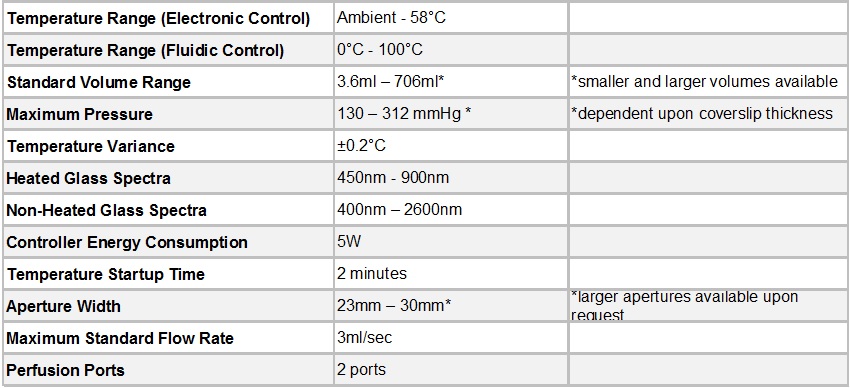
Specifications
Instructions
FCS2 Chamber Features
The Focht Chamber System 3 (FCS3 ®) is a closed system, live-cell micro-observation flowcell chamber that offers several advantages over other chambers. In addition to its unique perfusion and thermal control system, it is fully compatible with all modes of microscopy. It is also the only chamber to combine high-volume laminar flow perfusion rates with Koehler illumination and precise temperature control without an air curtain. Its user definable flow characteristics make it adaptable to nearly any protocol where cells are perfused in a flow-cell.
The FCS3 is a flow cell or flow chamber that is designed specifically for the demands of today’s live-cell imaging requirements. It has limitless flow characteristics because its flow geometry can easily be customized by the user. It can provide low shear near laminar flow as well as high shear in a directed flow and anything in between. The most important feature of the FCS3, other than unequalled temperature uniformity, is that there is a precise directed flow of media over the cells. Open dish type chambers do not provide the flow capabilities of the FCS3. The FCS3 provides an optical imaging cavity where the user can precisely define the optimum flow characteristics necessary for the experiment.
Specifications

Instructions
