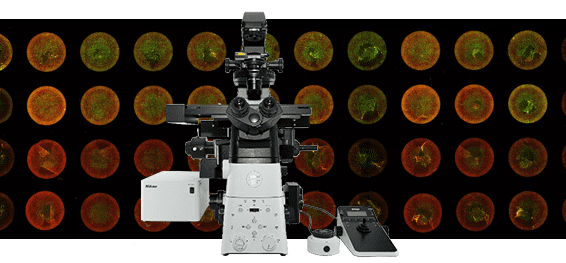

Nikon’s all-new inverted microscope platform for advanced imaging. See more than before!
The Eclipse Ti2 delivers an unparalleled 25mm field of view (FOV) that revolutionizes the way you see. With this incredible FOV, the Ti2 maximizes the sensor area of large-format CMOS cameras without making compromises, and significantly improves data throughput. The Ti2’s exceptionally stable, drift-free platform is designed to meet the demands of super-resolution imaging while its unique hardware-triggering capabilities enhance even the most challenging, high-speed imaging applications. Furthermore, the Ti2’s unique, intelligent functions guide users through imaging workflows by gathering data from internal sensors, eliminating the possibility of user errors. In addition, the status of each sensor is automatically recorded during acquisition, providing quality control for imaging experiments and enhancing data reproducibility. In combination with Nikon’s powerful acquisition and analysis software, NIS-Elements, the Ti2 is a total innovation in imaging.

As research trends evolve towards large-scale, systems-level approaches, there is an increasing demand for faster data acquisition and higher throughput capabilities. Development of large-format camera sensors and improvements in the data processing capabilities of PCs have facilitated such research trends. The Ti2, with its unprecedented 25mm field of view, provides the next level of scalability, enabling researchers to truly maximize the utility of large-format detectors and future-proof their core imaging platform as camera technologies continue to develop at a rapid pace.
High-power LEDs deliver bright illumination across the Ti2’s large field of view, ensuring clear, consistent results from demanding applications such as high-magnification DIC. Incorporation of a fly-eye lens design provides uniform illumination from edge to edge for quantitative high-speed imaging and seamless tiling of images in stitching applications.

A compact epi-fluorescence illuminator designed for large FOV imaging is equipped with a quartz fly-eye lens and provides high transmittance across a broad spectrum, including UV. Large diameter fluorescence filters with hard coatings deliver large FOV images with a high signal-to-noise ratio.

The diameter of the observation light path has been enlarged in order to achieve a field number of 25 at the imaging port. The resulting large FOV is capable of capturing approximately double the area of conventional optics, enabling users to gain maximum performance from large-format sensors such as CMOS detectors.

Objectives for large FOV imaging
Objectives with superior image flatness ensure high quality images from edge to edge. Utilizing the maximum potential of the OFN25 objective significantly accelerates data collection.
Nikon’s FX-format F-mount cameras Digital Sight 50M and Digital Sight 10 are equipped with CMOS image sensors optimized for research use, originally developed for professional D-SLR cameras. This allows high-speed and high-sensitivity live-cell imaging, enabling the best use to be made of the Ti2’s large FOV.

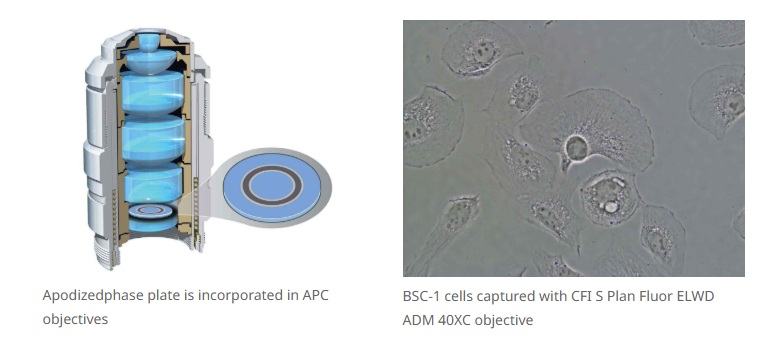
Nikon’s high-precision CFI60 infinity optics, designed for use with a variety of sophisticated observation methods, are highly regarded by researchers for their superb optical performance and solid reliability.
Nikon’s unique apodized phase contrast objectives with selective amplitude filters dramatically increase contrast and reduce halo artifacts to provide detailed high-definition images.
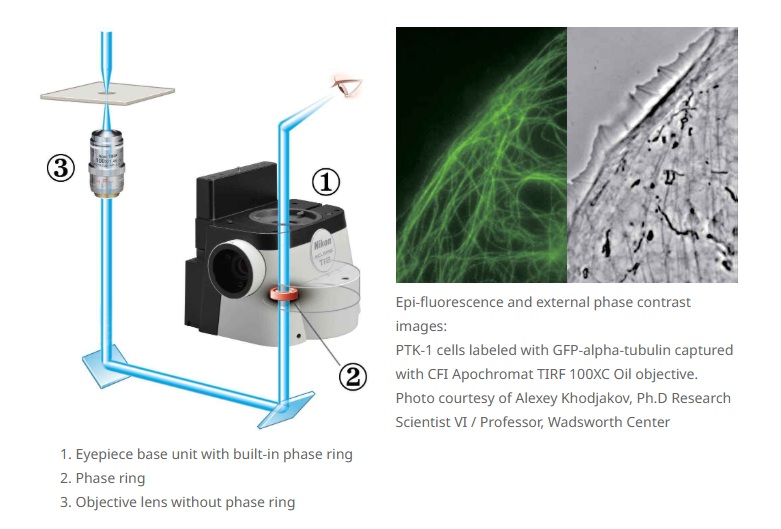
External phase contrast Ti2-E
The motorized external phase-contrast system enables users to combine phase contrast with epi-fluorescence imaging without compromising fluorescent light transmission by bypassing the need to use phase-contrast objectives. For example, very high NA, liquid immersion objectives can be used for phase-contrast imaging. Using this external phase contrast system, users can easily combine phase contrast with other imaging modalities, including weak-fluorescence imaging such as TIRF and laser tweezer applications.
Nikon’s highly-regarded DIC optics provide uniformly clear and detailed images with high resolution and contrast throughout the magnification range. DIC prisms are individually tailored for each objective lens to provide the highest-quality DIC images for every sample.

This is a plastic-compatible, high-contrast imaging technique for unstained, transparent samples such as oocytes. NAMC provides pseudo-three-dimensional images with a shadow-cast appearance. The direction of contrast can be easily adjusted for each sample.

Auto Correction Collar Ti2-E
Changes in sample thickness, cover glass thickness, refractive index distribution in the sample, and temperature can lead to spherical aberration and image deterioration. The highest quality objectives are often equipped with correction collars to compensate for these changes, and precise positioning of the collar is critical in achieving high resolution, high contrast images. This new auto correction collar utilizes a harmonic drive and automatic correction algorithm that enable users to easily achieve precise collar adjustment to achieve optimum objective performance every time.

Epi-fluorescence
The Lambda series objectives, utilizing Nikon’s proprietary Nano Crystal Coat technology, are perfect for demanding, low-signal, multi-channel fluorescence imaging that requires high transmission and aberration correction over a wide wavelength range. Combined with new fluorescence filter cubes that offer improved fluorescence detection and stray light countermeasures such as the Noise Terminator, the Lambda series objectives demonstrate their power in weak signal observations such as single-molecule imaging and even luminescence-based applications.

Volume Contrast
Volume Contrast technique utilizes a series of label-free, brightfield images captured at various Z-depths to assemble a phase distribution image.
Volume Contrast renders cells easily identifiable as objects for automated counting and area analysis. As this method utilizes brightfield imaging, Volume Contrast enables in-line, non-destructive analysis of cells, suitable for a wide variety applications. Note. For Ti2-E only.
 Brightfield Brightfield |
 Volume Contrast Volume Contrast |
HeLa cells imaged with CFI S Plan Fluor ELWD 20XC
Features of Volume Contrast(VC)
 Cell identification based on phase contrast images Cell identification based on phase contrast images
Cells encircled in red are incorrectly identified as a single object. |
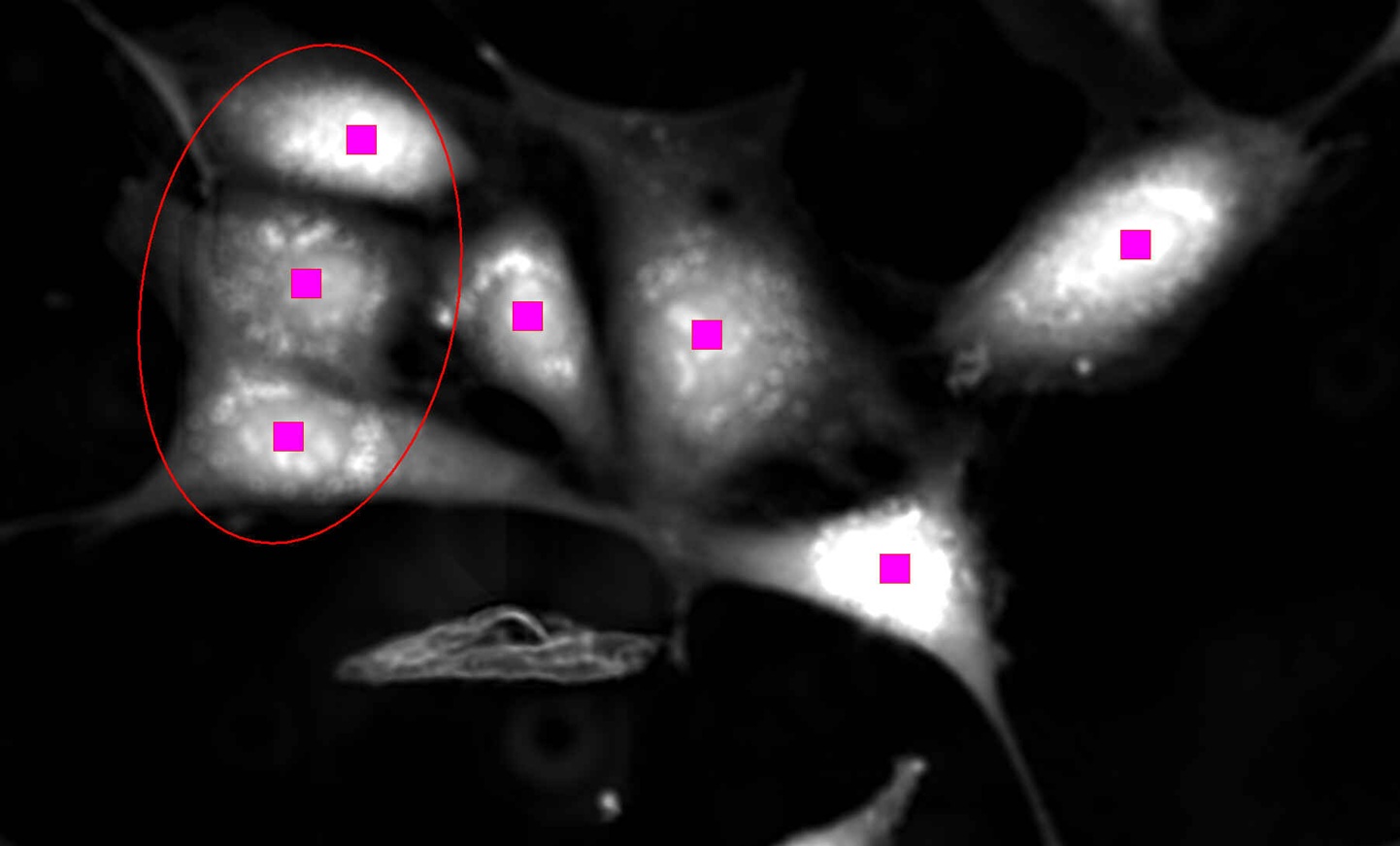 Cell identification using VC images Cell identification using VC images
Cells encircled in red are correctly identified as three separate objects, resulting in accurate cell counts. |
Phase contrast images are negatively impacted at the edges of wells due to the meniscus effect. Volume Contrast bypasses this effect and enables cells at the edges of wells to be clearly identified, resulting in increased cell counts and improved statistics.
 |
 VC image VC image |
Focus perfected
Even the slightest change in temperature and vibrations in the imaging environment can greatly impact focus stability. The Ti2 eliminates focus drift using both static and dynamic measures to enable faithful visualization of the nanoscopicand microscopic world during long time-lapse experiments.
Mechanically redesigned for ultra-high stability Ti2-E
In order to improve the focusing stability, both Z-drive and PFS autofocusing mechanisms have been completely re-designed.
The new Z-focusing mechanism is smaller and positioned adjacent to the nosepiece to minimize vibrations. It remains adjacent to the nosepiece even in an expanded (staged-up) configuration, ensuring stability for all applications.
The detector portion of the Perfect Focus System (PFS) has been detached from the nosepiece in order to reduce mechanical load on the objective nosepiece. This new design also minimizes heat transfer, which contributes to a more stable imaging environment. Towards this end, the power consumption of the Z-drive motor has also been reduced. Combined, these mechanical redesigns result in an ultra-stable imaging platform, perfectly suited for single-molecule imaging and super-resolution applications.
High stability Z-focusing mechanism remains adjacent to the nosepiece even in expanded configurations.① PFS nosepiece
② PFS Measuring Unit
The Perfect Focus System (PFS) automatically corrects focus drift caused by temperature changes and mechanical vibrations, which can be caused by a variety of factors including the addition of reagents to the sample and multi-position imaging.
The PFS maintains focus by detecting and tracking the position of the coverslip surface in real time. Unique optical offset technology allows users to easily maintain focus at a desired position offset from the cover slip surface. The PFS automatically and continuously maintains focus by means of a built-in linear encoder and high speed feedback mechanism, providing highly reliable images even during long-term, complex imaging tasks.
PFS is compatible with a wide range of applications, from routine experiments involving plastic culture dishes to single-molecule imaging and multi-photon imaging. It is also compatible with a wide range of wavelengths, from ultraviolet to infrared, meaning it can be used for multi-photon and optical tweezer applications.
The performance of long-term imaging using the PFS together with water immersion objectives can be increased by using the new Water Immersion Dispenser. The Water Immersion Dispenser automatically applies the appropriate amount of pure water to the tip of an objective, preventing the immersion liquid from drying out and overflowing during experiments. It is compatible with all types of water immersion objectives and helps to stably provide high-resolution, high-contrast and aberration-corrected time-lapse images over long periods of time.

It is no longer necessary to memorize complex microscope alignment and operation procedures. The Ti2 integrates data from sensors to guide you through these steps, eliminating user error and enabling researchers to concentrate on their data.
Built-in sensors detect the status of microscope components
A collection of built-in sensors detects and relays status information for a variety of components in the microscope. All of the status information is recorded in the metadata when you acquire images with a computer, so you can easily recall acquisition conditions and/or check for configuration errors.
In addition, a built-in internal camera allows users to view the back aperture, facilitating confirmation of phase ring alignment and extinction cross in DIC. It also provides a laser-safe method for aligning lasers for applications such as TIRF.
Microscope status can be viewed on a tablet and also determined based on status lights on the front of the microscope, enabling status determination in a dark room.
The Ti2’s Assist Guide function provides interactive step-by-step guidance for microscope operation. The Assist Guide can be viewed on a tablet or PC, and integrates real time data from built-in sensors and an internal camera. The Assist Guide is designed to help users through alignment procedures for both experiment setup and troubleshooting.
The Check Mode allows users to easily confirm, on either a tablet or PC that all the correct microscope components are in place for their chosen observation method. This capability eliminates time and effort normally required for troubleshooting when the desired observation method is not achieved. This functionality is particularly advantageous when multiple users are involved, each with the potential to make unexpected changes to the microscope settings. Custom check procedures can also be pre-programmed.
The Ti2 has been completely redesigned, from the overall body design to the selection and placement of every button and switch, for the ultimate in user experience. The controls are easy to use even in the dark, where the majority of imaging experiments are conducted. The Ti2 provides an intuitive and effortless user interface so researchers can focus on the data and not on microscope control.
The placement of all of the buttons and switches are based on the type of illumination they control. Buttons that control diascopicobservation are positioned on the left side of the microscope and those that control epi-fluorescence observation are on the right side. Buttons that control common operations are on the front panel. This use of zoning provides an easy-to-remember layout, a desirable feature when operating the microscope in a dark room.

Shuttle switches have been incorporated into the design to control devices such as the fluorescence filter turret and objective nosepiece. These types of switches emulate the feel of manually rotating these devices, for intuitive control. Additional functionality can be incorporated into these shuttle switches so that a single switch can operate multiple related devices. For example, the shuttle switch for the fluorescence filter turret not only rotates the turret but also opens and closes the fluorescence shutter when the user presses the switch. It is also possible to program these switches to operate a barrier filter wheel and the external phase contrast unit.
Conveniently located Function buttons allow customization of the user interface. Users can select from more than 100 functions, including control of motorized devices such as shutters and even signal output to external devices via the I/O port for triggered acquisition. Mode functions, which enable instant changing of observation methods by storing the settings of each motorized device, can also be assigned to these buttons.
A focus acceleration button and a PFS engagement button are provided adjacent to the focusing knobs. The two buttons are easily identified by touch because of their different shapes. Focusing speed is automatically adjusted for the objective in use, enabling stress-free operation by maintaining an ideal focusing speed.
The Ti2 joystick not only controls stage movement, but the majority of motorized functions on the microscope, including PFS activity. It can display XYZ coordinates and the status of microscope components, providing an effective means for the user to remotely control the microscope. Motorized functions of the Ti2 can also be controlled from a tablet, connected by wireless LAN to the microscope, providing a versatile graphical interface for microscope control.
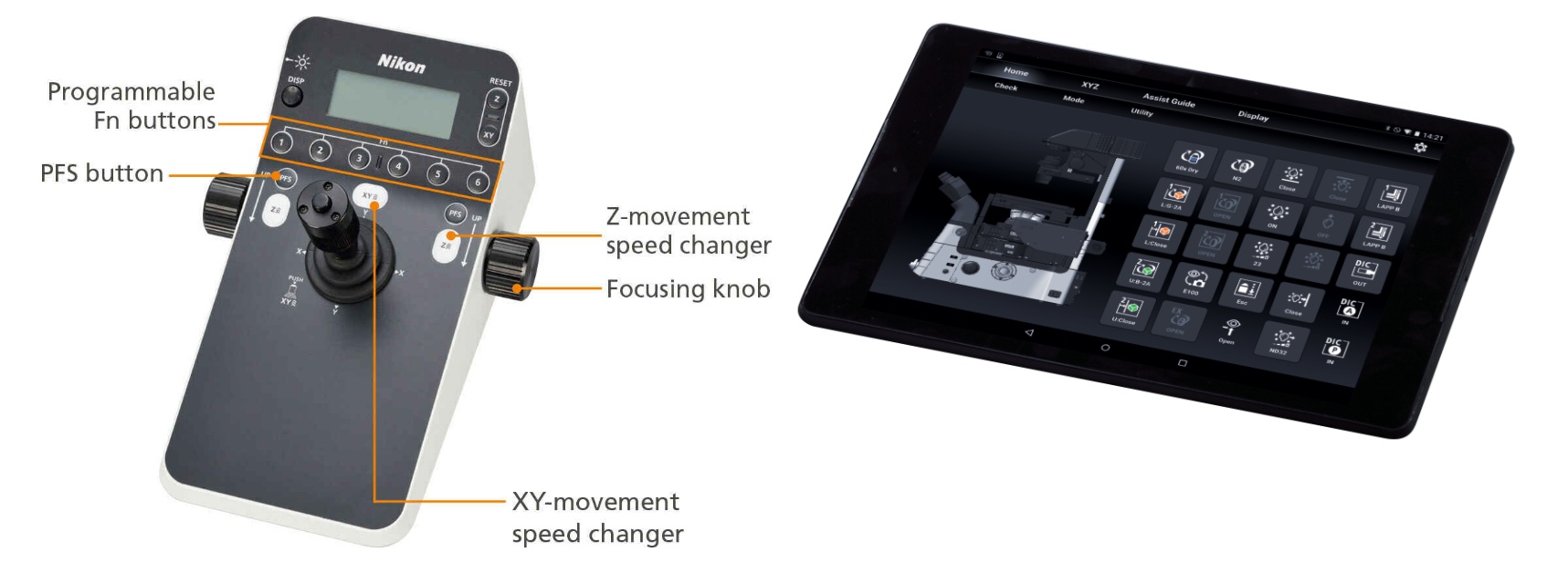

It enables setting and control of Ti2-E, and setting, status display and operational guidance of Ti2-A.
Ti2 Control can be downloaded for free from the App Store® and Google Play™.
Download it to a device such as an iPhone®, iPad® or Android™. See the “Operational conditions” section below for information about compatible devices.


iOS devices: devices equipped with iOS 10.3 or later (iPhone 7 or later, iPad Air 2 or later)
Android™ devices: devices equipped with Android 5.1 or later
| ECLIPSE Ti2-E/Ti2-E/B*1 | ECLIPSE Ti2-A | ECLIPSE Ti2-U | ||
|---|---|---|---|---|
| MAIN BODY | ||||
| Optical system | Infinity-corrected CFI60 | |||
| Field number*2 | 22 with C-mount, 25 with F-mount | |||
| Intermediate Magnification switching | Manual switching of 1.0x/1.5x (exchangeable from 1.5x to 2.0x) | |||
| Status detection | — | |||
| Bertrand lens | Manual in/out, and manual focus, Status detection | — | ||
| Output port | 4 Motorized positions
Eyepiece 100%, left 100%, right 100%, eyepiece 20%/left 80% (Ti2-E/B: bottom 100%) |
4 Manual positions
Eyepiece 100%, left 100%, right 100%, option (to eyepiece 20%/left 80% or eyepiece 20%/right 80%) |
||
| Can add ports by use of back port unit and/or choice of tube base unit*3 | ||||
| Focusing unit | Motorized drive, Coarse/fine focus changeover, 10mm stroke, Minimum increments: 0.01μm, 0.02μm (with encoder control) | Manual drive, Coarse/fine focusing knob, 10mm stroke | ||
| Stage up | Available*4 | |||
| TUBE BODY | ||||
| Binocular tube | Binocular S tube TC-T-TS (field number 22), Ergonomic ER tube TC-T-ER (field number 22) | |||
| Motorized eyepiece tube base unit for external PH (TI2-T-BP-E) | Camera port (field number 16), Motorized PH turret with 4 motorized positions | — | ||
| Assist eyepiece tube base unit (TI2-T-BA) | Assist camera (field number 22), Status detection | — | ||
| Eyepiece tube base unit with port (TI2-T-BC) | Camera port (field number 16) | — | Camera port (field number 16) | |
| TRANSMITTED ILLUMINATION | ||||
| Pillar for transmitted illumination (TI2-D-PD) | Condenser vertical stroke: 66mm, Backward tilting up to 25 degrees, With field diagram and refocusing mechanism 2 filter slot positions (4 filter position option is also available with Filter Slider for transmitted illumination (TI2-D-SF)) | |||
| LED Lamphouse for dia illumination (TI2-D-LHLED) | High power LED | |||
| Precentered Lamphouse (D-LH/LC) | 100W halogen bulb (pre-centered) | |||
| CONDENSER | ||||
| Motorized condenser turret (TI2-C-TC-E) | 7 motorized positions (ø37mm x4, ø39mm x3), LWD/ELWD/CLWD/NAMC condenser lenses are supported | — | ||
| Intelligent condenser turret (TI2-C-TC-I) | 7 manual positions (ø37mm x4, ø39mm x3), Status detection, LWD/ELWD/CLWD/NAMC condenser lenses are supported | — | ||
| Condenser turret (TC-C-TC) | 7 manual positions (ø37mm x4, ø39mm x3), LWD/ELWD/CLWD/NAMC condenser lenses are supported | |||
| ELWD-S condenser turret (TE-C) | 4 manual positions, With ELWD condenser lens (NA0.3/OD65) | |||
| HNA condenser slider (TI2-C-SCH) | 2 manual positions (ø37mm x1, ø39mm x1), HNA dry lens/HNA oil lens are supported | |||
| Condenser lens | LWD (W.D.=30mm, NA=0.52), ELWD (W.D.=75mm, NA=0.3), CLWD (W.D.=13mm, NA=0.72), HNA dry (W.D.=5mm, NA=0.85), HNA oil (W.D.=1.9mm, NA=1.3), NAMC (W.D.=44mm, NA=0.4) | |||
| STAGE | ||||
| Motorized stage (TI2-S-SE-E, TI2-S-SS-E) | Stroke X: ±57mm, Stroke Y: ±36.5mm, Max drive speed: approx. 25mm/sec, Magnetic sample holder | — | ||
| Stage (TC-S-SR, TC-S-SRF) | Stroke X: ±57mm, Stroke Y: ±36.5mm, Adjustable stroke range (3 levels) with adjusting pin, Long/middle/short handle options available | |||
| Gliding stage (TC-S-GS) | Stroke ø20mm | |||
| NOSEPIECE | ||||
| Perfect Focus Unit with motorized nosepiece for Auto Correction Collar (TI2-N-NDA-P) | 5 motorized positions, Simple waterproof structure | — | ||
| Motorized DIC sextuple nosepiece (TI2-N-ND-E) | Perfect Focus Unit with motorized nosepiece (TI2-N-ND-P)
Perfect Focus Unit with motorized nosepiece for MP (TI2-N-NDM-P) |
6 motorized positions, Simple waterproof structure | — | |
| Intelligent DIC sextuple nosepiece (TI2-N-ND-I) | 6 manual positions, Status detection, Simple waterproof structure | — | ||
| Sextuple nosepiece (TI2-N-N), DIC sextuple nosepiece (TI2-N-ND) | 6 manual positions, Simple waterproof structure | |||
| EPI-FILTER TURRET | ||||
| Motorized epi filter turret (TI2-F-FLT-E, TI2-F-FLTH-E) | 6 motorized positions, Motorized shutter | — | ||
| Intelligent epi filter turret (TI2-F-FLT-I) | 6 manual positions, Manual shutter, Status detection*5 | |||
| FILTER WHEEL/SHUTTER | ||||
| Motorized BA filter wheel (TI2-P-FWB-E) | 7 motorized positions, High speed mode: 50ms, Low vibration mode: 100ms (movement time between adjacent positions) | — | ||
| Motorized shutter (NI-SH-E)*6 | 12ms to open/close | |||
| EPI-FLUORESCENCE ATTACHMENT | ||||
| EPI-FL module (TI2-LA-FL), EPI-FL module for large FOV (TI2-LA-FLL) | Supports fiber illuminator; includes 2-position filter slider and aperture diaphragm | |||
| Simple EPI-FL attachment (TI2-F-FLS) | Supports both fiber illuminator and lamp house; includes 3-position filter slider | |||
| Field stop slider | Circular (TI2-F-FSC), rectangular (TI2-F-FSR), square (TI2-F-FSS) aperture options | |||
| CONTROL UNIT | ||||
| Controller, display device | Stage joystick (TI2-S-JS), Tablet | Tablet | — | |
| Controller for TI2-E (TI2-CTRE) | USB/LAN interface, I/O function | — | ||
| OPERATING ENVIRONMENT | ||||
| Temperature: 0℃+40℃, Humidity: 60% RH max. (at +40℃, no condensation), Indoor use only | ||||
Motorized accessories have a status detection function
*1 Motorized model with a bottom port
*2 Limitations apply based on objective and filter cube choice, stage-up configuration, and illumination module, etc.
*3 Tube base units with a port cannot be used with Ti2-A
*4 Stage up kit is required. Please contact Nikon.
*5 Status detection cannot be used when attached to the Ti2-U
*6 NI-SH-CON Controller for Motorized Shutter is required for use with the Ti2-A/Ti2-U